
Slow resorption
bone substitute

Obtained from
equine bone
thermal process:
the treatment that preserves
duration
Calcitos® is obtained through high temperature treatment of cancellous equine bone tissue, which displays substantial similarities with human bone at a morphological level and in terms of the chemical composition of bone apatite crystals.

The manufacturing process of Calcitos®
-
 SUPERIOR QUALITY RAW MATERIAL
SUPERIOR QUALITY RAW MATERIAL -
 THERMAL DEANTIGENATION
THERMAL DEANTIGENATION -
 BETA RAY STERILIZATION
BETA RAY STERILIZATION
THE TYPICAL CHARACTERISTICS
OF THERMAL TREATMENT,
WITH SOMETHING MORE
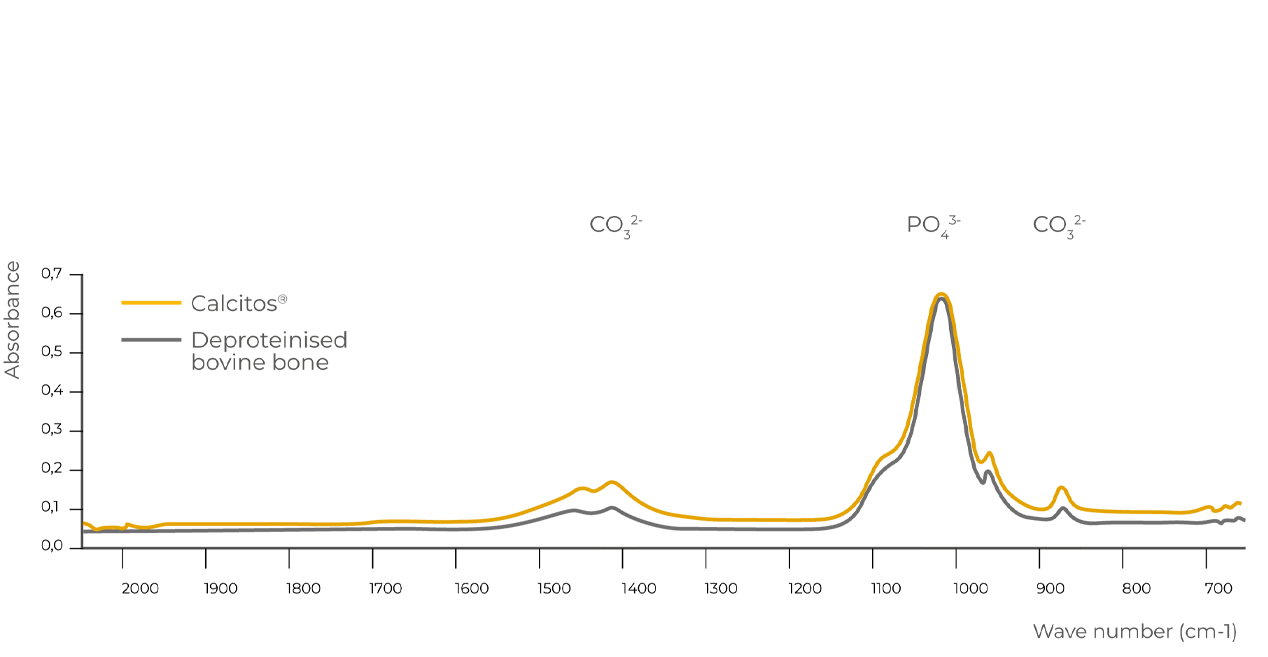
Calcitos® exhibits a mineral structure that is very similar to that of human bone and has the same chemical-physical properties of the other heterologous bone substitutes (xenografts) obtained through thermal treatment. The FT-IR infrared absorption spectrums of Calcitos® and of deproteinised bovine bone match, indicating that their chemical-physical composition is the same.
-
 THE HIGHEST
THE HIGHEST
LEVEL OF QUALITY -
 SAFETY OF THE
SAFETY OF THE
SPECIES OF ORIGIN
Versatile.
The ideal solution in every case
Calcitos® is indicated for all cases where the desired outcome is the continuing stability of regenerated bone volume over time. Its chemical-physical characteristics, the natural presence of macro- and micro-pores suitable for vascularization, cellular adhesion and hydration, make it a versatile bone substitute that acts as a scaffold for the placement of new bone tissue while also serving as a space keeper.
-
 MICRO-PORES AND MACRO-PORESThe natural structure of macro- and micro-pores that features the equine bone of which Calcitos® is made of guarantees excellent hydrophilicity, facilitates vascularization and enables the perfect integration of the material into the newly-formed bone.
MICRO-PORES AND MACRO-PORESThe natural structure of macro- and micro-pores that features the equine bone of which Calcitos® is made of guarantees excellent hydrophilicity, facilitates vascularization and enables the perfect integration of the material into the newly-formed bone. -
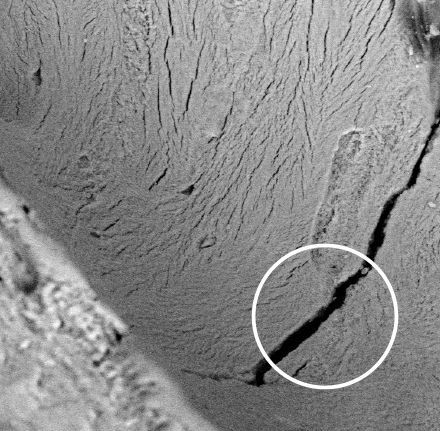 ELIMINATION OF COLLAGENThe thermal treatment totally removes the organic component, including bone collagen1. The bone crystallizes, and the fracture points display characteristic fissures.
ELIMINATION OF COLLAGENThe thermal treatment totally removes the organic component, including bone collagen1. The bone crystallizes, and the fracture points display characteristic fissures. -
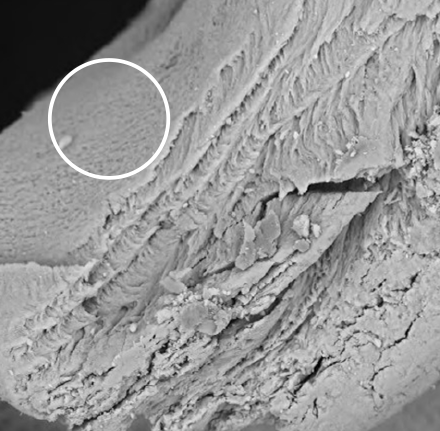 CALCINED SURFACEThe high temperatures applied modify the surface structure of the bone. The osteoblasts come together to form new vital bone tissue, while the degradative action of the osteoclasts is reduced2: favoring slow resorption.
CALCINED SURFACEThe high temperatures applied modify the surface structure of the bone. The osteoblasts come together to form new vital bone tissue, while the degradative action of the osteoclasts is reduced2: favoring slow resorption.
APPLICATIONS
Every situation has its optimum protection
-


IDEAL
PROTECTION
TIMEFOR PREDICTABLE BONE
REGENERATION:- Resorbable natural pericardium
- Protection time 3-4 months
- Suturable
- Traction resistant
- Ideal for the majority of surgical applications.
-


SPECIFIC
PROTECTION
TIMEFLEX CORTICAL SHEET
TO MAINTAIN BONE PROFILES IN AESTHETIC AREA- Flexible cortical bone
- Protection time > 6 months
- It remodels completely with new vital bone
- Restores cortical profiles and keeps them stable over time.
Product range

Calcitos®
Cancellous granules
Cancellous granules 0,25-1 mm
| OMC-029n | Cancellous Granules – 1 btl / 0.25g 0.25-1 mm |
| OMC-030n | Cancellous Granules – 1 btl / 0.5g 0.25-1 mm |
| OMC-031n | Cancellous Granules – 1 btl / 1g 0.25-1 mm |

Heart®
Membrane made of pericardium
Pericardium Membrane
| HRT-001 | Pericardium membrane – 1 pc. 25 x 30 x 0.2-0.4 mm |
| HRT-002 | Pericardium membrane – 1 pc 50 x 30 x 0.2-0.4 mm |
| HRT-003n | Pericardium membrane – 1 pc 15 x 20 x 0.2-0.4 mm |
| HRT-005n | Pericardium membrane – 1 pc 20 x 20 x 0.2-0.4 mm |

Osteoxenon®– Osteoplant® *
Flex Cortical Sheet
Flex Cortical Sheet
| OSP-OX03 | Cortical Membrane – 1 pc. 25 x 25 x 0.2 mm |
| OSP-OX04 | Cortical Membrane – 1 pc. 50 x 25 x 0.2 mm |
| OSP-OX08 | Cortical Sheet- 1 pc 20-25 x 30-35 x 0.9 mm |
| OSP-OX09 | Cortical Sheet- 1 pc 20-25 x 30-35 x 0.5 mm |
| OTC-C2 | Cortical Sheet – 1 pc 20-25 x 30-35 x 0.9 mm |
| OTC-C3 | Cortical Sheet- 1 pc 20-25 x 30-35 x 0.5 mm |
| OTC-CE | Cortical Membrane – 1 pc. 25 x 25 x 0.2 mm |
| OTC-CE2 | Cortical Membrane – 1 pc. 50 x 25 x 0.2 mm |
![]()
* the availability of Flex Cortical Sheet in the Osteoxenon® or Osteoplant® range varies from country to country.


